Key Takeaways
- The FDA approved Foundayo (orforglipron) in April 2026 as the first oral GLP-1 weight loss medication that can be taken with food at any time of day.[1]
- In phase 3 trials, orforglipron produced 12.4% weight loss (27.3 lbs) at 72 weeks — less than injectable tirzepatide (20%) but comparable to oral semaglutide.[2][3]
- GLP-1 medications reduced major adverse cardiovascular events by 20% in the SELECT trial, with benefits extending beyond weight loss alone.[4]
- Without continued treatment, most patients regain 60–80% of lost weight within 1–2 years.[5]
- Self-pay pricing starts at $149/month for Foundayo; covered insurance can bring the cost to as low as $25/month.[6]
- A boxed warning exists for thyroid C-cell tumors; these drugs are contraindicated in anyone with personal or family history of medullary thyroid cancer.[1]

The Weight Loss Medication Revolution
A decade ago, the pharmacologic options for obesity were few and underwhelming. Available drugs produced, on average, about 5% body weight reduction — meaningful for metabolic health, but nowhere near what patients hoped for. That conversation has changed. GLP-1 receptor agonists routinely produce 15–20% weight loss in clinical trials, rivaling results previously seen only with bariatric surgery.[2]
More than 12% of U.S. adults have now used a GLP-1 medication, and prescriptions continue to climb. Patients who struggled with obesity for decades are seeing sustainable results for the first time.
The catch? Until 2026, almost every effective option required weekly injections. That created three problems. First, needle avoidance — a meaningful fraction of patients refused injectable therapy entirely. Second, manufacturing: injectable pens require sterile fill-finish capacity that simply can't scale quickly, and patients waited months in shortages. Third, the oral option that did exist — Rybelsus, later reformulated as an oral Wegovy pill — required taking the pill on an empty stomach with strict fasting afterward.[3]
Foundayo (orforglipron), approved by the FDA on April 1, 2026, changes that calculus. It is the first truly food-flexible oral GLP-1 approved for weight loss. This guide walks through how these drugs work, which patients benefit, what the trial data actually show, what to expect in terms of side effects and cost, and the difficult question of what happens when you stop.
What GLP-1 Medications Actually Do
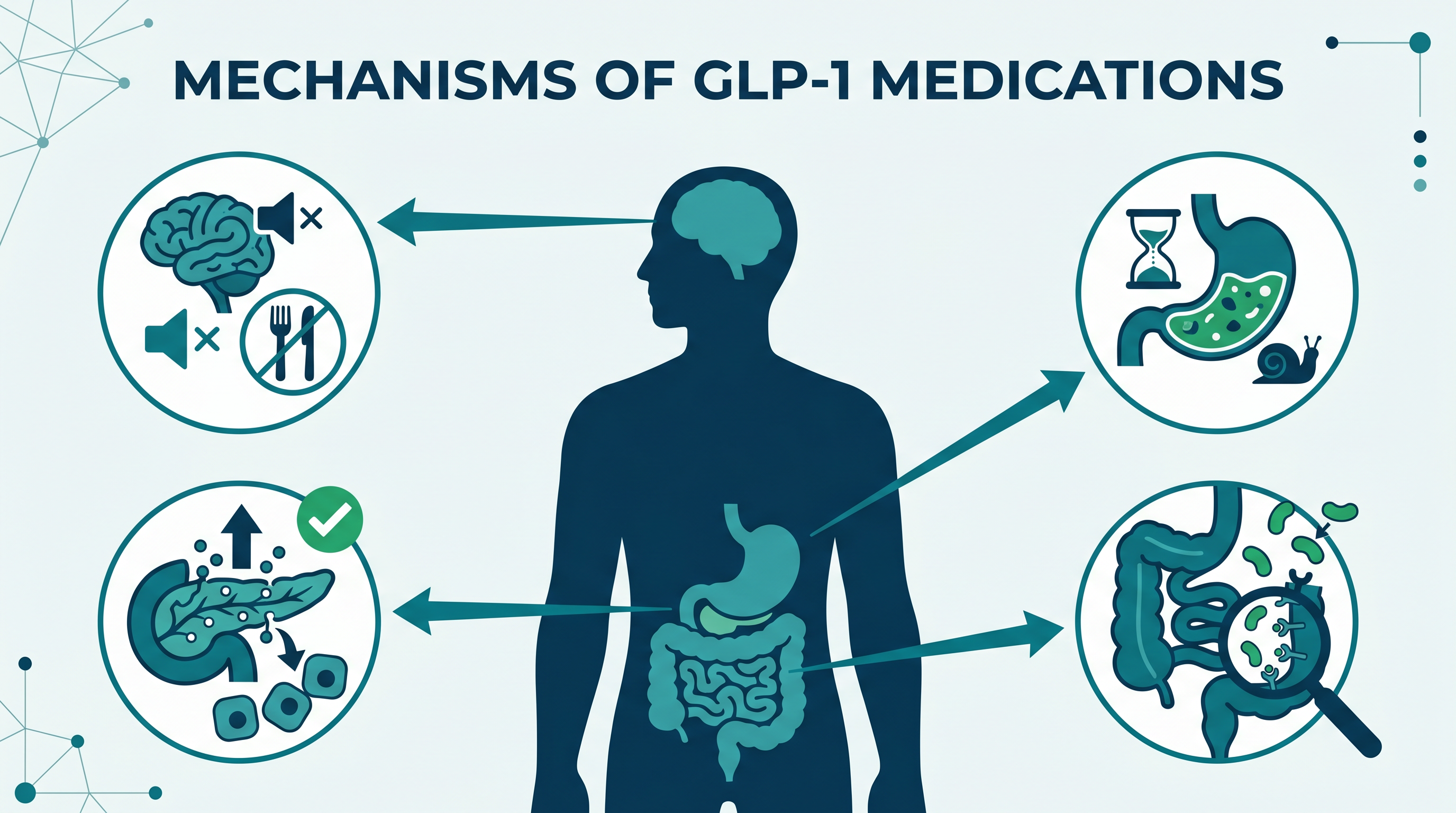
GLP-1 — glucagon-like peptide-1 — is a hormone your gut already makes. After you eat, your intestinal L-cells release GLP-1, which signals to your brain, pancreas, and digestive tract that food has arrived. The problem is that natural GLP-1 breaks down within minutes. GLP-1 receptor agonists are synthetic molecules engineered to mimic the hormone while resisting breakdown, so the signal lasts hours or days instead.
Four effects explain most of what these drugs do:
1. The brain: quieting "food noise." GLP-1 receptors in the hypothalamus and reward centers reduce appetite and the intrusive thoughts about food that many patients with obesity describe. A common refrain in survey and qualitative data is some variation of "I just stopped thinking about food all day." That phenomenon — often called food noise reduction — is one of the most consistent subjective effects of the class.
2. The stomach: delayed gastric emptying. GLP-1s slow how quickly the stomach releases food into the small intestine. You feel full faster and stay full longer. This is also why nausea is such a common early side effect — the slower emptying can feel uncomfortable while the body adjusts.
3. The pancreas: glucose-dependent insulin release. When blood sugar rises after a meal, GLP-1 amplifies the pancreas's insulin response. When blood sugar is normal, it doesn't. That glucose-dependent action is why GLP-1s rarely cause hypoglycemia unless combined with insulin or sulfonylureas.
4. The gut: satiety signaling. Receptors throughout the intestine reinforce the sense of fullness and dampen the feedback loops that drive "just one more bite."
Put together, these effects add up to roughly 300–500 fewer calories eaten per day — without the willpower battle that usually accompanies dieting. The drug isn't burning fat; it's making undereating feel natural.
The GLP-1 Medications Available in 2026
The class has expanded quickly. Here's what you'll actually see prescribed for weight loss or weight-related indications in 2026:
| Brand | Generic | Form | Weight Loss (trials) | Primary Use |
|---|---|---|---|---|
| Foundayo | orforglipron | Oral pill daily (no food restriction) | 12.4% at 72 weeks[2] | Weight loss |
| Wegovy (oral) | oral semaglutide | Oral pill daily (empty stomach, 30-min fast) | ~14% in ACHIEVE-3 comparator arm[3] | Weight loss |
| Wegovy | semaglutide 2.4 mg | Weekly injection | 14.9% (STEP-1)[5] | Weight loss |
| Ozempic | semaglutide | Weekly injection | Used off-label for weight | Type 2 diabetes |
| Zepbound | tirzepatide | Weekly injection | 20.2% (SURMOUNT-1)[7] | Weight loss |
| Mounjaro | tirzepatide | Weekly injection | Off-label weight use | Type 2 diabetes |
| Saxenda | liraglutide | Daily injection | 5–6% | Weight loss |
A few things worth noting. Tirzepatide (Zepbound/Mounjaro) is a dual GLP-1/GIP agonist, which explains its edge on weight loss. Semaglutide and orforglipron are pure GLP-1 agonists. Liraglutide has largely been eclipsed by the newer agents, though it remains available and inexpensive.
The real-world choice usually comes down to three questions: pill or injection? Do you have type 2 diabetes? What does your insurance cover? Patients without diabetes seeking maximum weight loss typically do best on tirzepatide. Patients who will not inject often end up on orforglipron. Patients with diabetes frequently get dual benefit from semaglutide or tirzepatide.
Foundayo (Orforglipron): The New Oral Option

Orforglipron's April 2026 approval is notable for several reasons. It used the FDA's new Commissioner's National Priority Voucher (CNPV) pathway — a faster review track — and became the first new molecular entity approved under that program.[1]
Orforglipron is also chemically different from previous oral GLP-1s. Rybelsus and the oral Wegovy pill are peptides — essentially the same injectable molecule wrapped with absorption enhancers. That's why they need an empty stomach and a 30-minute fast afterward. Orforglipron is a small non-peptide molecule, absorbed the way a typical pill is absorbed. No fasting. No water restriction. No specific time of day.
The ATTAIN-1 trial tested orforglipron in 3,127 adults with obesity or overweight with a weight-related condition. At 72 weeks on the top 36 mg dose, patients lost an average of 12.4% of body weight — about 27.3 pounds — compared with 0.9% in the placebo group. Nearly 60% of participants lost at least 10% of their body weight, and 39.6% lost at least 15%.[2]
The head-to-head ACHIEVE-3 trial, published in The Lancet in February 2026, compared orforglipron 36 mg to oral semaglutide 14 mg in 1,698 patients with type 2 diabetes over 52 weeks. Orforglipron won on both endpoints: A1C dropped 2.2% versus 1.4% with oral semaglutide, and weight loss was 9.2% versus 5.3%. That's roughly 74% greater relative weight loss with the oral orforglipron.[3]
Dosing follows a monthly escalation schedule designed to minimize GI side effects: 0.8 mg → 2.5 mg → 5.5 mg → 9 mg → 14.5 mg → 17.2 mg, with the target maintenance dose reached after about six months. The schedule is deliberately slow. Trial data consistently show that patients who rush escalation have more nausea and vomiting, while those who stay on each step for the full month tolerate the drug better.
Orforglipron doesn't match tirzepatide's 20% weight loss. But it beats or matches every other oral option, comes without fasting restrictions, and manufacturing is simpler — which should translate into more reliable supply. For patients who won't inject, for patients in regions where the cold chain for injectables is unreliable, and for first-time GLP-1 users who want a lower-commitment starting point, it's an appealing choice.
Cardiovascular and Other Benefits
Weight loss gets the headlines, but the broader benefits of this class are arguably more important. The SELECT trial, published in 2023, enrolled 17,604 adults with overweight or obesity and established cardiovascular disease — but without diabetes. Over an average of about three years, semaglutide 2.4 mg reduced major adverse cardiovascular events (cardiovascular death, non-fatal heart attack, non-fatal stroke) by 20% compared to placebo.[4]
What makes SELECT remarkable is the mechanism. Analyses suggest that only about a third of the cardiovascular benefit is explained by weight loss alone. The other two-thirds appears to come from direct effects on inflammation, vascular function, and blood pressure. That positions GLP-1s as disease-modifying therapy, not just weight loss drugs.
In trials, GLP-1 medications also reduce:
- Non-HDL cholesterol and triglycerides
- Systolic blood pressure (typically 3–6 mmHg)
- High-sensitivity C-reactive protein (hsCRP), a marker of systemic inflammation — by as much as 47.7% in ATTAIN-1[2]
- HbA1c in patients with type 2 diabetes or prediabetes
Early and emerging evidence suggests benefits in other conditions too. Semaglutide improved non-alcoholic steatohepatitis (NASH) in a phase 3 trial. Tirzepatide reduced the severity of obstructive sleep apnea meaningfully in SURMOUNT-OSA. Early signals point to kidney protection in patients with chronic kidney disease and overweight. These indications are being actively studied, and additional FDA approvals for non-weight-loss uses are anticipated over the next few years.
What this adds up to clinically: GLP-1s are best understood as treating the underlying metabolic disease, not just as a weight loss intervention. That framing matches what patients report — blood pressure improving, energy returning, knees hurting less.
Who Should Consider GLP-1 Therapy
FDA indications for weight loss require:
- BMI ≥ 30 (obesity), or
- BMI ≥ 27 (overweight) with at least one weight-related condition — hypertension, type 2 diabetes, dyslipidemia, obstructive sleep apnea, cardiovascular disease, or similar
These thresholds set eligibility, but they don't answer the more important question: who benefits most? The strongest evidence-based case for GLP-1 therapy is in patients who have both overweight or obesity and established cardiovascular disease. SELECT showed that group gets significant MACE reduction, which is a mortality-relevant outcome. Second strongest: patients with type 2 diabetes, where semaglutide or tirzepatide treats both conditions in one prescription.
Other clear candidates include patients with BMI 35+ who have tried and failed multiple structured weight loss approaches, patients with obesity plus severe sleep apnea, and patients with a metabolic syndrome phenotype where reducing insulin resistance matters. Every patient in these groups deserves a real conversation about whether GLP-1 therapy fits their values and goals.
A point worth emphasizing: these medications are not a substitute for lifestyle work. They work alongside nutrition and physical activity. Without adequate protein intake and resistance training, a significant share of the weight lost on a GLP-1 will be lean mass rather than fat — particularly concerning for older adults where sarcopenia is already a risk.
Off-label use for low-BMI cosmetic weight loss is not supported by the evidence, exposes the patient to real risks (including rapid muscle loss and rebound after discontinuation), and typically isn't covered by insurance. This use is outside FDA-approved indications.
Who Should NOT Take GLP-1s
- Personal or family history of medullary thyroid carcinoma (MTC). This is the boxed warning. Rodent studies showed thyroid C-cell tumors with GLP-1 exposure. Whether this translates to humans is debated, but the contraindication is strict.[1]
- Multiple Endocrine Neoplasia syndrome type 2 (MEN2) — a genetic condition that predisposes to MTC.
- Prior serious hypersensitivity to a GLP-1 medication.
Beyond those absolute contraindications, there are situations that call for caution or alternative approaches:[8]
- History of pancreatitis. GLP-1s have been associated with acute pancreatitis. Clinical guidance generally advises avoiding this class in patients with a pancreatitis history unless there's no reasonable alternative.
- Severe gastrointestinal disease — gastroparesis, severe reflux, or uncontrolled IBD. Slowing gastric emptying can worsen these conditions.
- Active or unstable diabetic retinopathy. Rapid improvement in glucose control has been associated with transient worsening of retinopathy, which has been observed in GLP-1 trials.
- History of gallbladder disease. Rapid weight loss increases gallstone risk.
- Pregnancy or planning pregnancy. These medications are not recommended in pregnancy. Manufacturers generally recommend stopping at least two months before trying to conceive.
- Upcoming general anesthesia. Delayed gastric emptying increases aspiration risk during anesthesia. Most anesthesiologists now ask patients to hold GLP-1s for 1 week (daily drugs) or 2 weeks (weekly injections) before elective surgery.
- On another GLP-1. You cannot combine two GLP-1 medications. Stacking is unsafe and not evidence-based.
Side Effects: What to Expect
GI side effects dominate the profile. In the ATTAIN-1 trial of orforglipron, the most common adverse events were nausea (21–33% depending on dose), constipation (21–29%), diarrhea (21–23%), vomiting (13–24%), and heartburn. About 10.3% of patients on the 36 mg dose stopped the drug because of side effects, compared with 2.6% on placebo.[2] Other GLP-1s have similar profiles.
Two things matter about these numbers. First, rates are highest during dose escalation and usually improve with time on a steady dose. Second, they are manageable with a few practical strategies that clinicians commonly advise before starting therapy:
- Stay on each escalation step for the full recommended interval. Don't let enthusiasm push you up faster. Patients who take their time almost always do better.
- Eat smaller, more frequent meals. Large meals against a slowly emptying stomach are a recipe for nausea. Clinicians often advise stopping at about 75% of what feels like "full."
- Prioritize protein — 25–30 grams at each meal, aiming for at least 1.0–1.2 g per kg of body weight daily. This preserves muscle mass during weight loss.
- Hydrate deliberately. Reduced thirst is common; dehydration makes nausea worse and raises kidney injury risk.
- Avoid high-fat and very sweet foods early on — they are the most common reported triggers for nausea and vomiting in clinical observation.
Serious side effects are rare but real. Pancreatitis presents as severe, persistent upper abdominal pain often radiating to the back, and it requires you to stop the drug immediately and seek evaluation. Acute kidney injury can happen when dehydration from vomiting or diarrhea is left untreated. Gallbladder disease — stones, cholecystitis — is increased during rapid weight loss. Hair loss is reported but usually reflects the shock of rapid weight change and resolves over months.
Muscle mass loss deserves specific attention. Studies suggest 25–40% of total weight lost on a GLP-1 comes from lean body mass in patients who don't strength train. The concern is greatest in older patients, where functional capacity and fall risk are at stake. The fix isn't medical — it's resistance training two to three times a week plus adequate protein. That counseling is as important as the prescription itself.
The Weight Regain Question
This is one of the hardest conversations in obesity medicine, and it's one that should happen before starting therapy, not after. The weight lost on these medications does not stay off once the medication stops. That statement is as well-supported as any other finding in the field.
The STEP-1 extension trial followed patients who had lost weight on semaglutide and then switched to placebo. Within one year, participants had regained roughly two-thirds of the weight they had lost.[5] The SURMOUNT-4 trial showed similar patterns for tirzepatide: patients switched from the drug to placebo regained about 14% of body weight over the following year, effectively erasing most of their loss.[7] An Oxford analysis looking at real-world patients found that average time to return to pre-treatment weight after discontinuation was about 1.7 years.[5]
Real-world adherence data reinforce the problem. About 50% of patients discontinue GLP-1 therapy within one year, and roughly 70% stop within two years — usually because of cost, side effects, or a belief that they can maintain results without the medication.[5] Most of those who stop regain the weight.
Why does this happen? Because GLP-1 medications treat the biology of obesity, they don't cure it. The metabolic adaptations that drive weight regain — lower resting energy expenditure, elevated hunger hormones, increased reward response to food — return when the drug is removed. This is identical to how blood pressure medications work: they control the disease as long as you take them.
Two things are worth keeping in mind. First: plan for long-term, possibly lifelong therapy. If you're not ready for that commitment, a GLP-1 may not be the right fit right now. Second: if you do eventually need to come off, taper slowly with your physician's guidance, double down on protein and resistance training, and expect that some regain is likely. A few maintenance strategies are in early research — including preclinical work on agents like TIX100 designed specifically for weight maintenance after GLP-1 — but none are clinically available yet.
Cost and Access in 2026
Price is the single biggest barrier for most patients. Here's how it breaks down in 2026:[6]
| Medication | Self-Pay (monthly) | Insurance / Savings Card |
|---|---|---|
| Foundayo (orforglipron) | $149 (starting dose) → $299–349 (maintenance) | As low as $25/month with commercial insurance + Lilly Savings Card; $50/month via Medicare GLP-1 Bridge (through Dec 2026) |
| Wegovy (oral semaglutide) | $149–299/month | $25/month with commercial insurance + Novo Nordisk savings programs |
| Wegovy (injectable 2.4 mg) | $199–449/month direct-to-patient | $25–75/month with coverage + savings card |
| Zepbound (tirzepatide) | $349–499/month via LillyDirect | $25/month with commercial insurance + savings card |
A few things to know about coverage. Most commercial insurance plans require prior authorization for GLP-1s used for weight loss, and most demand documentation of BMI plus a prior attempt at structured weight management. Some plans limit coverage to 12 months. Medicare has historically not covered weight-loss-only indications, though the 2026 GLP-1 Bridge program provides temporary access to Foundayo at $50/month through December.
Direct-to-consumer pharmacies like LillyDirect and telehealth platforms like Ro have made self-pay access simpler than it used to be. For patients paying out of pocket, the difference between $149 and $449 a month comes from the specific drug, dose, and delivery pathway — not from differences in quality.
A warning about compounded semaglutide: the FDA does not approve compounded versions of GLP-1s, and the agency has issued specific warnings about safety risks in 2026. With manufacturing of approved products now keeping up with demand, the prior justification for compounded GLP-1s has largely disappeared. Current clinical guidance strongly discourages their use. On April 30, 2026, the FDA proposed to exclude semaglutide, tirzepatide, and liraglutide from the 503B bulks list entirely, citing no clinical need for large-scale compounding now that FDA-approved supply is stable.
What This Means for You
If you're considering a GLP-1, here's the practical roadmap a physician is likely to walk you through:
Start with a physician visit. You need an actual evaluation — BMI calculation, review of weight-related conditions, medication history, and screening for contraindications. Telehealth visits are well-suited for this; the initial evaluation, lab review, dose titration, and monthly check-ins can all be done remotely.
Baseline labs are standard. Most physicians will order a complete blood count (CBC), a metabolic panel (CMP), lipid panel, HbA1c, and TSH before starting. Some add lipase. These establish a baseline and screen for conditions — thyroid disease, kidney issues, undiagnosed diabetes — that affect drug choice.
Set realistic expectations. Weight loss on a GLP-1 typically starts in the first month, picks up during dose escalation, and plateaus around 12–14 months. The final weight loss depends on the drug, the dose, and how well lifestyle factors support it. Expect 10–15% on orforglipron, 14–15% on injectable semaglutide, and 18–20% on tirzepatide — if you take the drug consistently at target dose with lifestyle support.
Plan for long-term therapy. Before you start, think through whether you're prepared for a multi-year or indefinite commitment. If not, a different approach may be a better starting point.
Invest in muscle preservation. Resistance training two to three times per week and 1.0–1.2 g/kg of daily protein are not optional — they are part of the treatment. Without them, a meaningful fraction of your weight loss will be muscle.
Expect monthly contact with your physician during titration. Dose adjustments, side effect management, and lab follow-up are the norm. Once you're stable on a maintenance dose, follow-up can stretch to every 3 months.
Red Flags: When to Contact Your Doctor
- Severe or persistent abdominal pain, especially upper abdominal pain radiating to the back — this can signal pancreatitis and requires immediate evaluation.
- Vomiting that prevents you from keeping fluids down — this raises the risk of dehydration and kidney injury.
- Signs of dehydration — dizziness when standing, reduced urine output, dark urine, or confusion.
- Rapid or racing heart that isn't explained by exertion or anxiety.
- A new neck lump, hoarseness, or voice changes — these can be signs of thyroid involvement and warrant evaluation.
- Pregnancy — stop the medication and contact your physician immediately.
- Before any surgery or procedure with anesthesia — inform your surgical and anesthesia teams that you are on a GLP-1; most will ask you to hold the drug for 1–2 weeks beforehand.
- Severe right upper abdominal pain with nausea — possible gallbladder disease.
Frequently Asked Questions
Most patients need long-term or lifelong therapy. Trial data show that stopping a GLP-1 leads to regain of roughly two-thirds of lost weight within a year.[5] Obesity is a chronic disease, and GLP-1s treat it the way blood pressure medications treat hypertension — the benefit lasts only as long as the treatment. Some patients taper to a lower maintenance dose with their physician's guidance.
Foundayo can be taken at any time of day with or without food, which simplifies drug interactions. However, because GLP-1s slow gastric emptying, they can affect absorption of some oral medications. If you take medications with a narrow therapeutic window — such as levothyroxine, warfarin, or some seizure medications — your physician may monitor levels more closely. Tell your prescriber about every medication you take, including over-the-counter products and supplements.
For a daily oral GLP-1 like Foundayo, take the missed dose as soon as you remember — unless it is close to your next scheduled dose. Do not double up. For weekly injectables like Wegovy or Zepbound, you can take the dose within 5 days of your normal schedule; if more than 5 days have passed, skip that dose and resume your weekly schedule. Missing several doses in a row may require restarting at a lower dose to avoid reintroducing GI side effects.
No. The FDA does not approve compounded versions of semaglutide or tirzepatide and has issued warnings about them in 2026. Compounded formulations are not evaluated for safety, purity, or potency, and dosing errors have caused serious adverse events. With the 2026 expansion of Foundayo and Wegovy production, the prior shortages that drove compounded use are largely resolved. Current clinical guidance strongly discourages compounded GLP-1 use.
Coverage varies widely. Many commercial plans now cover GLP-1s for weight loss with a prior authorization that requires documented BMI criteria and evidence of a prior weight loss attempt. Medicare historically did not cover weight-loss-only indications, but the 2026 GLP-1 Bridge program allows Medicare patients $50/month access to Foundayo through December 2026.[6] If you have diabetes, injectable semaglutide or tirzepatide for that indication is usually covered. Call your insurer before you start to confirm specifics.
Stop the medication and contact your physician immediately. GLP-1s are not recommended during pregnancy — animal studies have shown fetal harm, and human data are limited. Most manufacturers recommend discontinuing the drug at least two months before a planned pregnancy because of the long elimination time of some agents. GLP-1s can also restore fertility in some women who previously had irregular ovulation, so reliable contraception matters if pregnancy is not planned.
References
- U.S. Food and Drug Administration. FDA Approves First New Molecular Entity Under National Priority Voucher Program. Press announcement, April 2026. https://www.fda.gov/news-events/press-announcements/fda-approves-first-new-molecular-entity-under-national-priority-voucher-program
- Eli Lilly and Company. Lilly's oral GLP-1, orforglipron, delivers weight loss of up to an average of 27.3 lbs in first of two pivotal phase 3 trials in adults with obesity (ATTAIN-1). 2025. https://www.prnewswire.com/news-releases/lillys-oral-glp-1--orforglipron-delivers-weight-loss-of-up-to-an-average-of-27-3-lbs-in-first-of-two-pivotal-phase-3-trials-in-adults-with-obesity-302523649.html
- Scientific American. Head-to-Head Trial Shows Eli Lilly's Oral GLP-1 Orforglipron Outperforms Oral Semaglutide (ACHIEVE-3, The Lancet). February 2026. https://www.scientificamerican.com/article/head-to-head-trial-shows-eli-lillys-oral-glp-1-orforglipron-outperforms-oral/
- American College of Cardiology. SELECT: Semaglutide Reduces Risk of MACE in Adults with Overweight or Obesity. https://www.acc.org/latest-in-cardiology/articles/2023/08/10/14/29/select-semaglutide-reduces-risk-of-mace-in-adults-with-overweight-or-obesity
- MedCentral. Weight Maintenance After GLP-1 RA Withdrawal Exposes Critical Research Gaps. https://www.medcentral.com/endocrinology/obesity/weight-maintenance-after-glp-1-ra-withdrawal-exposes-critical-research-gaps
- Ro. Foundayo Cost: What to Expect in 2026. https://ro.co/weight-loss/foundayo-cost/
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide Once Weekly for the Treatment of Obesity (SURMOUNT-1). New England Journal of Medicine. 2022;387(3):205-216. https://www.nejm.org/doi/full/10.1056/NEJMoa2206038
- GoodRx. Who Should Not Take GLP-1 Agonists? Contraindications and Precautions. https://www.goodrx.com/classes/glp-1-agonists/who-should-not-take-glp-1
- Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP-1). New England Journal of Medicine. 2021;384(11):989-1002. https://www.nejm.org/doi/full/10.1056/NEJMoa2032183
- American Diabetes Association. Standards of Care in Diabetes — 2026: Obesity and Weight Management for the Prevention and Treatment of Type 2 Diabetes. https://diabetesjournals.org/care/issue/49/Supplement_1
- Centers for Disease Control and Prevention. Adult Obesity Facts. https://www.cdc.gov/obesity/adult-obesity-facts/index.html